The classes that met today were given time to begin work on a 'Make-up HW' assignment, where you turn in all HW that you have not done on time throughout the year. Do NOT turn this assignment in one piece at a time; all of this work is to be stapled together and turned in during the first few days we are back in school (starting Oct. 17).
Please use common sense and realize that you must complete all of the assignments for a given week, in order to get the check minus makeup credit for that week.
Tuesday, September 27, 2011
Monday, September 26, 2011
HW#7a -- Monday, Sept. 26
Write down the electron configuration for these elements:
(Remember to include every orbital that you cross, on the way to get to the element!)
1) Zinc
2) Sulfur
3) Bromine
4) Cadmium
For example, let's do nitrogen. You'll need the HALF SHEET elements table for this, as it shows you where the different orbitals (s, p, d and f) are.
Start in teh top left box (hydrogen, or "H") and count every electron that you pass as you move in a line to the right. I first pass two electrons in the 1s section, then two electrons in the 2s section, and finally three in 2p. Since we've now arrived at and counted the electron for the symbol "N," we're done. You would write the result like this:
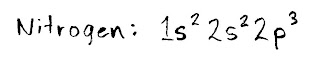
(Remember to include every orbital that you cross, on the way to get to the element!)
1) Zinc
2) Sulfur
3) Bromine
4) Cadmium
For example, let's do nitrogen. You'll need the HALF SHEET elements table for this, as it shows you where the different orbitals (s, p, d and f) are.
Start in teh top left box (hydrogen, or "H") and count every electron that you pass as you move in a line to the right. I first pass two electrons in the 1s section, then two electrons in the 2s section, and finally three in 2p. Since we've now arrived at and counted the electron for the symbol "N," we're done. You would write the result like this:
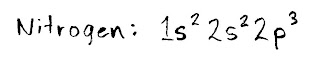
Friday, September 23, 2011
Thursday, September 22, 2011
Wednesday, September 21, 2011
HW#6c -- Wednesday, Sept. 21
For HW on this day, finish problems #1-6 on the 'Bohr model' worksheet that we started on today in class. You are welcome to try the last two or three, marked with stars, for bonus credit. Remember, there are three rules:
1) Fill in the energy levels closest to the nucleus first
2) Do not move up to the next energy level until you've filled the one before it (1st ring fills with two electrons, 2nd ring fills with eight, 3rd ring fills with eight, etc.)
3) Only use exactly as many electrons as you have. For example, Boron's atomic number is 5, meaning that it will have five protons and, being neutral, it will have exactly five electrons.
You may find this online periodic table (click here) helpful for completing this exercise, and it might also help you finish coloring, labelling and writing two facts about each of the families on your periodic table.
1) Fill in the energy levels closest to the nucleus first
2) Do not move up to the next energy level until you've filled the one before it (1st ring fills with two electrons, 2nd ring fills with eight, 3rd ring fills with eight, etc.)
3) Only use exactly as many electrons as you have. For example, Boron's atomic number is 5, meaning that it will have five protons and, being neutral, it will have exactly five electrons.
You may find this online periodic table (click here) helpful for completing this exercise, and it might also help you finish coloring, labelling and writing two facts about each of the families on your periodic table.
Tuesday, September 20, 2011
HW#6b -- Tuesday, Sept. 20
Your homework for today is to finish labelling with color the seven element families on the period table. In the end, your table should have:
Alkali Metals
Alkaline-Earth Metals
Halogens
Noble Gases
Transition Metals
Lanthanides
Actinides
You should write the name of each family on the front, next to the family. Also, you need to have two facts written down for each family (can be on the front or the back).
If you're not going to use the textbooks in our classroom to finish working on this (i.e., during lunch, after school), then you can read about the families here (click for link). Just click each different link at the bottom left to read about the different families.
Keep in mind that there are still three more families close to where the 'staircase' is on the table (by silicon, for example) that we will talk about later.
Alkali Metals
Alkaline-Earth Metals
Halogens
Noble Gases
Transition Metals
Lanthanides
Actinides
You should write the name of each family on the front, next to the family. Also, you need to have two facts written down for each family (can be on the front or the back).
If you're not going to use the textbooks in our classroom to finish working on this (i.e., during lunch, after school), then you can read about the families here (click for link). Just click each different link at the bottom left to read about the different families.
Keep in mind that there are still three more families close to where the 'staircase' is on the table (by silicon, for example) that we will talk about later.
Monday, September 19, 2011
HW#6a -- Monday, Sept. 19
Check out this site (click for link) for an online version of the periodic table that will help you complete this assignment.


Subscribe to:
Comments (Atom)





